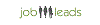Stipendio medio: euro73.333 /annuale
Più statisticheRisultati della ricerca: 9 offerte di lavoro
For a Pharmaceutical company, we are looking for
ASSOCIATE SCIENTIST - CHEMISTRY
Specific for the role
• Executes scientific experiments under limited supervision contributing to the conception, design, and interpretation of scientific data in support of research...
...Site Name: Italy - Siena
Posted Date: Apr 18 2024
Associate Scientist Protein Biochemistry (Fixed Term - 12 months contract)
Protein Biochemistry Function in Bacterial Vaccines R&D Italy and Global Quality organization
We are looking for a:
An Associate...
...Site Name: Italy - Siena
Posted Date: Apr 19 2024
Associate Scientist (Maternity Replacement) – GSK Institute for Global Health
Job purpose
As Associate Scientist you will:
Executes scientific experiments under limited supervision contributing to...
Job Title: CLINICAL RESEARCH ASSOCIATE
Location: Siena - Italy
Department: Clinical
Philogen S.p.A. would like to hire a CLINICAL RESEARCH ASSOCIATEfor the development of clinical experimental activity. The successful candidate will have responsibility for the management...
...Site Name: Italy - Siena
Posted Date: Mar 27 2024
Associate Director, Analytical Separation Sciences – Vaccines R&D
Are you excited about taking on a dynamic, high-impact role that involves leading a team of experts (10-15 direct reports based in Italy), collaborating...
...HospitalityWe believe that true hospitality springs from the nurturing and building of strong and lasting relationships with fellow associates, guests, partners, and the communities. We do this by recognizing and valuing each other on a personal level, understanding...
...the clinical activity.
~Perform documentation control in the TMF and quality reviews of clinical trial essential documents and associated documentation (including Protocols, Investigator’s Brochure, CRFs, etc.) as appropriate.
~Perform routine internal System and TMF...
...objectives and may be recognised as a key player in external international pharmacovigilance activities (i.e., may be a member of trade association or external initiatives).
Recognised as an authority with expert knowledge of relevant pharmacovigilance regulations and...
...and designers.
Independently handling situations and issues related to data quality and the configuration/access of resources associated with regulated production
Participating in the management of change requests in the client's IT/OT infrastructure
Operating...