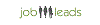Stipendio medio: euro37.500 /annuale
Più statisticheRisultati della ricerca: 7 offerte di lavoro
35.000 € - 40.000 €
Il mio cliente è azienda che sviluppa e produce dispositivi medici di classe IIa, IIb e III. Per loro conto siamo alla ricerca di una risorsa da inserire nel dipartimento Affari Regolaotori che si occuperà di gestire le attività di registrazione in ambito UE, FDA e altri...
...Mechanical Engineering, Single Use Devices, as well as other cross-functional teams like human factors, UI, quality, regulatory, PMO and medical affairs. In the end, the leader is responsible to further strengthen and continue our journey of world-class embedded software...
...through education, alignment, and cross-functional internal and external collaboration.
Partner with Corporate Legal Regulatory, Government Affairs, and government agencies to influence and advocate for or against international trade legislation with impact to Mozarc...
...documentation (requirements, standard checklists, risk analysis);
Collaborate with other R&D teams and with other departments (Quality, Regulatory, Clinical, Marketing);
Plan and manage project tasks according to the timeline and estimate effort.
Must Have :
Master...
...Design for Six Sigma or related product development processes (e.g. DOE, Montecarlo Analysis)
Familiarity with medical device regulatory environment including FDA regulations and guidance such as QSR, GMP, Usability
Familiarity with intellectual property and patent...
...workshops to promote awareness and understanding of CSV compliance
Support and participate in Group IT Quality-related audits (regulatory audits):
Collaborate with audit teams to prepare for and participate in quality-related audits.
Assist in gathering and...
...software components into medical devices.
Ensure that all software development activities are properly documented in accordance with regulatory requirements and company procedures.
Support upper management to review progress and allocate budgets for software development...